Patient and Public Involvement and Engagement
What is patient and public involvement and engagement?
Patient and Public Involvement and Engagement, or PPIE, is the processes of researchers collaborating with patients, caregivers, family members, service users and members of the public during research activities. INVOLVE defines public involvement as “research being carried out 'with' or 'by' members of the public rather than 'to', 'about' or 'for' them.
Involvement may include, helping to develop patient information leaflets, joining advisory and steering groups, or acting as co-applicants on research projects.
Participation may involve participating in a clinical trial or completing a questionnaire as a part of a research study.
Why is PPIE important to research?
It is vital to collaborate with members of the public to ensure that our research is of the highest quality, and focuses on issues which patients, and the public, find most important. By collaborating with members of the public and patients, our research will have a greater focus and relevance for those directly affected by the findings.
At the Bristol Centre for Surgical Research, we actively engage with the public and encourage their involvement in the work we do.
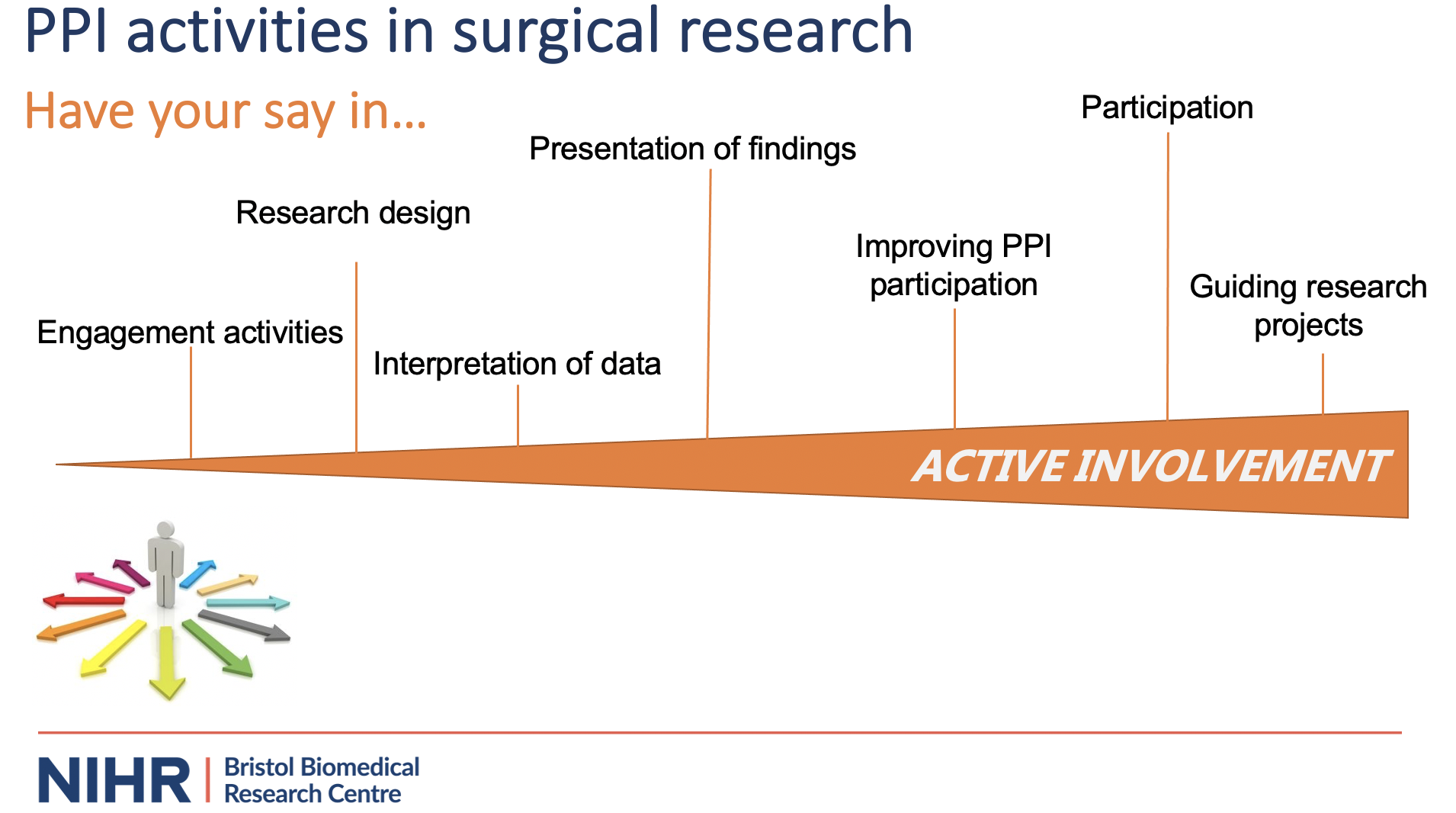
Our PPIEP activities
To deliver successful studies, members of the public are involved in a whole range of research activities that include study design, conduct, and the way research findings are shared.
Our main public involvement group meets regularly to discuss and collaborate on research activities within the National Institute of Health Research (NIHR) Bristol Biomedical Research Centre (BRC), which is linked to the Centre.
Our approach to public involvement in research maps to NIHR’s vision of involving patients and the public in research. Further information and guidance about public involvement in research can be found here
Patient and public contributions in our research
Many of our individual studies also have their own specific public involvement groups. For example, in the ROMIO (Randomised Oesophagectomy: Minimally Invasive or Open) feasibility study, public contributors helped to plan and secure funding for the study as well contributing to the design of patient-facing documents (such as study information leaflets). Patients and members of the public also helped to explore methods to improve patient questionnaire completion, some PPI members are co-authors on study publications and have helped to gain funding for a subsequent main study.
